Matter in Our Surroundings MCQ / Objective questions Chapter 1 Class 9 Science
What will be the volume of water in the beaker if we add one teaspoon of salt to it?
- a. Increase
- b. Decrease
- c. No change
What will happen to volume if we keep adding salt to the beaker in the above question?
- a. Increase
- b. Decrease
- c. No. change
- d. Negligible increase
In which conditions solubility of a substance will increase?
- a. Increase in temperature.
- b. Increase in pressure.
- c. Decrease in temperature
- d. None
Heating the bottom of the steel pot filled with water heats the water at the top also. This happens due to:
- a. Conduction
- b. Convection
- c. Radiation
- d. None
Which among these will have no shape but a fixed volume
- a. An aluminum pot.
- b. An iron chisel.
- c. Mustard oil
- d. Plastic bottle
Heat transfer from one terminal of a long iron rod to another is due to:
- a. Conduction
- b. Convection
- c. Radiation
- d. None
Which among these will have maximum kinetic energy?
- a. Solid
- b. Liquid
- c. Gas
- d. All have the same kinetic energy.
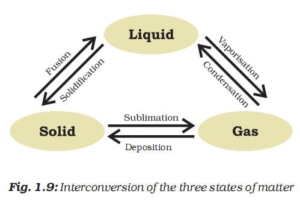
Heating ice converts ice into water. This energy is constant and is equal to
- a. 224 joule/gm
- b. 334 joule/gm
- c. 2230 joule/gm
- d. 2434 joule/gm
Which among these will be a more complicated mishap
- a. Burn due to spillage of hot water
- b. Spillage of boiling water
- c. Exposure of heated steam at 100ºc
- d. None
Which process causes the wind to blow?
- a. Conduction
- b. Convection
- c. Radiation
- d. None
Which among these will have the highest force of intermolecular attraction?
- a. An iron rod
- b. 100 ml of water
- c. Carbon dioxide produced by a fire extinguisher
- d. All three have an equal intermolecular force of attraction.
See also:
- Diversity in Living Organisms MCQ.
- Natural Resources MCQ.
- Tissues Class 9 MCQ
- The Fundamental Unit of Life MCQ
- Why do we fall ill MCQ
Matter in Our Surroundings MCQ / Objective questions Chapter 1 Class 9 Science
Ref: Chapter 1.