NCERT Science class 9 chapter 2 Is Matter Around Us Pure Exercise Answer and solution
1. Which separation techniques will you apply for the separation of the following?
(a) Sodium chloride from its solution in water.
(b) Ammonium chloride from a mixture containing sodium
chloride and ammonium chloride.
(c) Small pieces of metal in the engine oil of a car.
(d) Different pigments from an extract of flower petals.
(e) Butter from curd.
(f) Oil from water.
(g) Tea leaves from tea.
(h) Iron pins from sand.
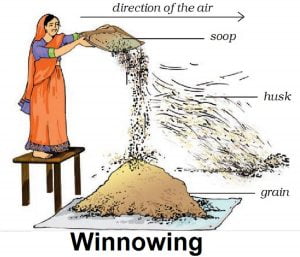
(i) Wheat grains from husk.
(j) Fine mud particles suspended in water.
Answer:
a) Evaporation: To separate salt ( sodium chloride) we will use evaporation. By evaporation, water molecules will vaporise and go away while salt will deposit at the bottom.
Note: We use the same method to get salt from the sea. We put salt water of the ocean into big open spaces. The energy of sunlight dries the mixture and salt deposit at the bottom.
b) Sublimation: Camphor, naphthalene, ammonium chloride sublimate (solid to gas ) easily on heating. We will boil the solution. We will get ammonium chloride by cooling the vapours, while salt will deposit at the bottom.
c) Filtration: Metals are insoluble in engine oil. We can easily filter them out using a filter or sieve.
d) Chromatography: Pigments of a flower petal have varying solubility in water. A chromatograph paper will separate the pigments.
e) Centrifugation: Butter is lighter than the curd. Centrifugation will separate butter from the curd and butter will start floating on the top.
f) Separating funnel: Oil is immiscible to water as one does not dissolve in others. Oils are lighter than the water, as a result, in separating funnel water will sink at the bottom while oil will float on water.
g) Filtration: We will use a tea-strainer to separate tea leaves from tea.
h) Magnetic Separation: Magnets attract iron but not the sand. When will bring a magnet to the mixture, iron will stick to the magnet while sand will remain as such?
j) Centrifugation, Evaporation.
3. Solubility table question 3.